Can Foreign Medical Service Advertise In Usa
Promoting Medical Devices Prior to FDA Approving or Clearance*
Posted on October 19, 2015 by
three min read
Near med device companies will agree with Tom Petty: the waiting is the hardest part. The time period from FDA submission to clearance or approval can feel like an eternity. Only what tin companies do to get hospitals and providers excited about their engineering science earlier launch?
A device that lacks FDA marketing clearance is considered "investigational," and therefore the company cannot promote, advertise or accept orders for it. The FDA considers such activities to be "commercialization of an unapproved device." The FDA does make some exceptions for trade shows, yet, then let us pause information technology down for yous.
Devices with Awaiting 510(g) Submissions
510(k) submissions are used to grant marketing approval for devices that have demonstrated the same effectiveness and safety as some other largely equivalent, legally marketed device. Which devices require a 510(k) submission? Any Class I, Ii or Iii device to exist marketed in the U.S. that:
- is intended for human employ,
- does non require Premarket Approval application (PMA),
- is not "exempt from 510(one thousand) requirements of the Federal Food, Drug and Cosmetic Deed (FD&C Act)," and
- does not "exceed the limitations of exemptions in .ix of the device regulation capacity."
The FDA doesn't permit companies to market devices with awaiting 510(yard) submissions. However, some devices are considered exempt from 501(k) submissions: these devices were legally marketed prior to May 28, 1976 and are termed "pre-amendment" devices. Importantly, to remain exempt from 501(one thousand) submissions, these devices must:
- have the same intended use,
- not be modified,
- non have a published FDA regulation requiring a PMA, and
- be endemic by the same company that owned the device prior to May 28, 1976.
Boosted devices are besides exempt from 501(k) submissions; click here to learn more almost these special circumstances.
Devices with IDE Blessing
If we're talking about an investigational device studied under an approved IDE application, you lot cannot say that it'south safety and effective for investigational utilize or promote it until afterwards the FDA has approved it for commercial distribution. However, in the guidance "Preparing Notification of Availability of Investigational Medical Devices and for Recruiting Study Subjects" (issued 1985 and revised 1999), the FDA clarified that y'all can publicize the availability of an investigational device with the purpose of recruiting clinical investigators for clinical trials. See how they did that? Simply make certain yous denote the device's availability in a medical or scientific publication or at a medical or scientific conference and clearly state that the purpose is but to obtain investigators and non to brand the device generally bachelor.
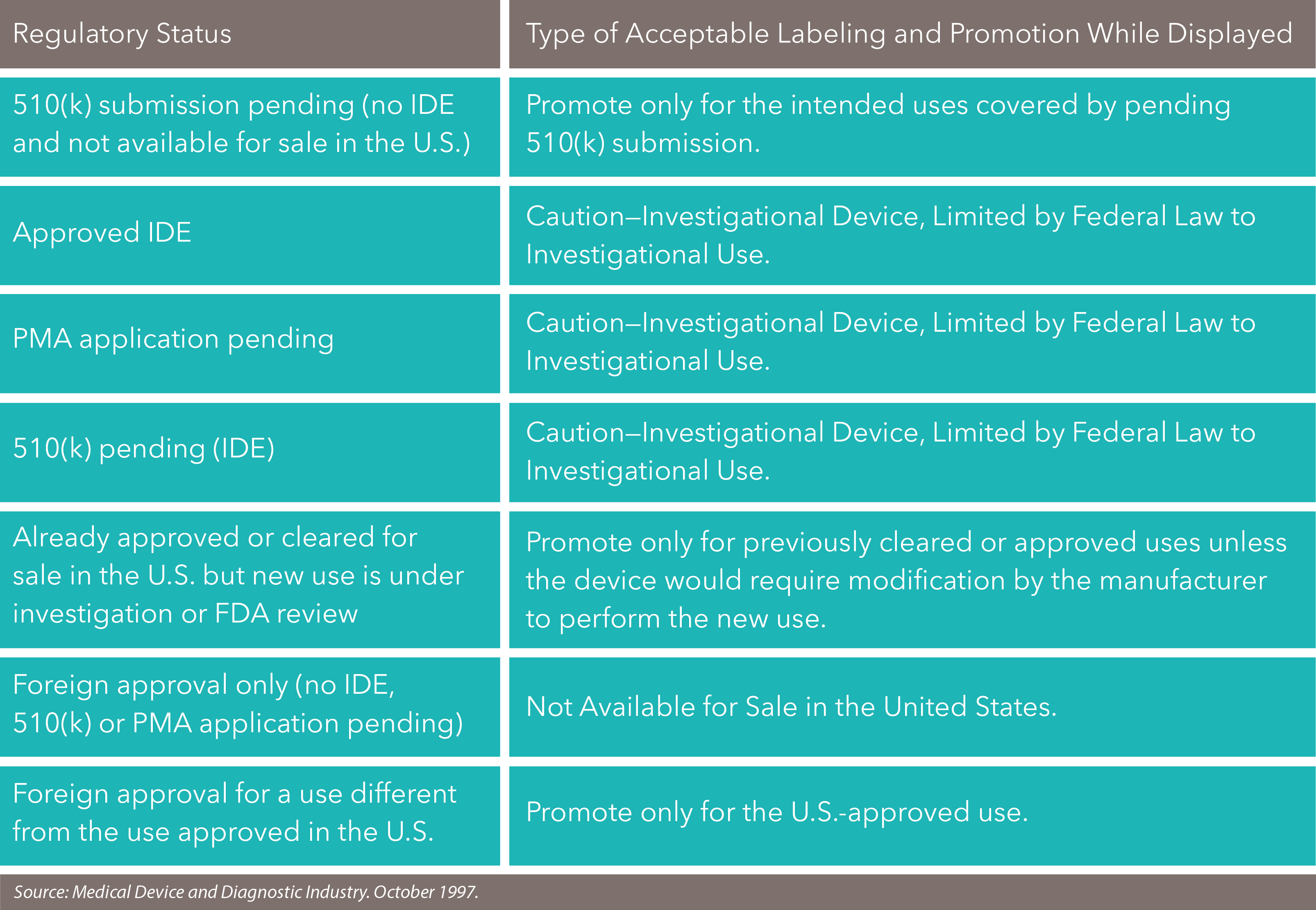
The FDA will frown upon mass mailings to solicit clinical investigators. And enrolling more investigators than necessary to evaluate the prophylactic and effectiveness of the device is also considered bad form. You can't make claims or imply that your investigational product is reliable, durable, rubber, or effective for the purposes under investigation. And no one likes a humble brag, peculiarly the FDA, so nothing any superiority claims or comparative descriptions.
The FDA mostly accepts that products displayed while a PMA application is pending goes beyond investigator recruitment. The safest course is to follow the same guidelines for investigational devices and keep off topics similar pricing, POs, and prospects for approval. A company tin talk about features and how the device operates.
To embrace your bases, y'all should take a sign (and say during an oral presentation): "Caution: Investigational Device. Express by Federal (or United States) Law to Investigational Utilise." Or if it's pending a PMA, say "Pending PMA, not available for sale within the U.s.a..".
Nearly FDA-canonical Devices
If your product is virtually approved (i.east., discipline of an approvable alphabetic character from the FDA), the FDA volition review whether the company can promote the device on a case-by-instance basis. In certain circumstances, a company may exist permitted to train physicians on the new device before the official clearance or approval is in hand.
The FDA besides has pretty strict rules about coloring within the lines. If a device has received 510(k) clearance or premarket approval, you lot need to stay inside the parameters of its canonical uses. The FDA disapproves of you talking or promoting your production for investigational or unapproved new uses. Because the device is already available, the FDA fears that you may be encouraging off-label use, which is a no-no.
While the devil is always in the detail when the FDA is involved, device manufactures that follow these guidelines should remain in the FDA's good graces when displaying an investigational or unapproved device at a trade show.
* This cursory outline should not be relied on equally legal advice, but every bit general background to assist in understanding certain FDA promotion and advertisement problems.
Need Marketing Help?
Grey Affair Marketing is a full-service, award-winning PR and marketing agency working exclusively with healthcare companies. We deliver the expertise and experience so clients tin make meliorate concern decisions and see better business organization results. Contact us and allow us aid you create more effective outreach strategies during this unprecedented time.
Can Foreign Medical Service Advertise In Usa,
Source: https://www.greymattermarketing.com/blog/promoting-medical-devices-prior-to-fda-approval-or-clearance
Posted by: boydreste1954.blogspot.com


0 Response to "Can Foreign Medical Service Advertise In Usa"
Post a Comment